Hi all! Sorry for the week delay - conferences and moving apartments. In my last piece, I took a 30,000 foot view of Apria Healthcare and how this 25-year DME titan has withstood the test of time, while still being plagued by the same issues as the rest of the industry.
We’ve talked about a bit about how the DME system works under normal conditions, but what happens when something breaks? Well, this week, I’m covering the ongoing Philips Respironics recall - this is a situation affecting more than 4 million oxygen-related devices, with an estimated 2 million in the US alone.
For those who don’t know, Philips Respironics is one of the world’s leading oxygen-based equipment manufacturers. Just to clarify, a DME manufacturer is usually a different entity than the DME supplier - Philips Respironics does not usually sell equipment directly to individuals. Instead, they work with a contracted network of DME suppliers to handle individual orders and reimbursement, as is the US industry standard. While the manufacturers don’t negotiate reimbursement rates with payors like DME providers do, these manufacturers still have to convince various health include their equipment on formularies.
Oxygen DME
Oxygen-based DME treats a number of well known conditions, from asthma to sleep apnea to symptoms of COVID-19. Even if you’re not super aware of this category, you probably remember the 2020 hospital ventilator crisis re COVID-19, especially in NYC - that’s oxygen-based DME. Another major area of oxygen DME are used for the various DME treatments for Obstructive Sleep Apnea (OSA) - a condition where a patient has difficulty breathing during sleep cycles. OSA DME include Bilevel Continuous Pressure Air Pumps (BiPAPs) and Continuous Pressure Air Pumps (CPAPs), which both simulate breathing through continuous air pressure (biPAPs have different levels of pressure for inhale and exhale).
OSA DME can be quite expensive, ranging from $300-$800 for a standard machine, and up to $3,000 when factoring in special features and accessories. To manage cost, many payors require their eligible members to rent from suppliers rather than buy outright, with a rental payments capped at 13 months, and total payment not to exceed the price of purchase, at which time the ownership title will transfer from the DME supplier to the patient. Payors also require suppliers to do monthly check-ins on patients to make sure they are actually using the devices they have purchased. In cases where the supplier determines and documents inadequate usage, the supplier repossesses the DME from the patient. This area can be quite lucrative for suppliers given the absolute dollar values the understandably increased admin and regulatory burden required keeps many DME suppliers away from this category altogether.
The Recall
On June 14 of this year, Philips Respironics announced a voluntary recall for more than 15 device types - from CPAPs and BiPAPs to ventilators - that were manufactured between 2009 and April of 2021. “Voluntary” recalls are not acts of kindness by manufacturers - these are risk management measures that manufacturers take because the alternative is the FDA taking control of the wheel and effectuating a mandatory recall, which is an outcome that neither the manufacturer nor the FDA prefers.
Anyways, Philips (allegedly in 2020) discovered that the polyester-based polyurethane (PE-PUR) sound abatement foam contained in many of their oxygen models presented a carcinogenic risk if ingested. Sound abatement foam itself is not uncommon across manufacturers, as it reduces the operational volume of these machines, but once it was discovered that this particular foam could come apart, especially when exposed to ozone cleaning material or high heat, and that this material present in 4M+ devices was in fact a cancer risk, Philips rightly took steps for an immediate recall.
So yeah, this was no joke: the FDA deemed the Philips move a Class I recall (the most severe): “a situation in which there is a reasonable probability that the use of or exposure to a violative product will cause serious adverse health consequences or death.” Even so, Philips had to vary their guidance based on product. For recalled ventilators, Philips advised patients to make no changes until they could find an adequate replacement. After all, a material contingent literally couldn’t breathe without these machines - discontinuing ventilators in the short term was not an option. In the case of CPAPs and BiPAPs, Philips recommended that patients immediately discontinue all usage and work with their physicians to determine next steps, as the discontinuation of these machines are considered unlikely to cause death or serious harm, even if it would reduce quality of life. Philips also began operating a recall site where patients could register their Philips device to determine their replacement eligibility and to receive the latest updates for replacement/rework instruction as they became known.
Stakeholder Responses
Well, if this recall process had gone / was going well, I wouldn’t be writing a piece about it. For one, the only company that really needed to “make things right” for patients was Philips - but Philips couldn’t just do that with a snap of their finger. In September, months after the initial recall announcement, Philips finally got FDA approval for its rework plan of the Dreamscape 1 (the majority of the affected devices). The company estimates that it will be able to get through the reworks of the DreamScape by September 2022 - not exactly an easily digestible timeline for your average patient.
I’m not saying Philips is totally at fault here for these recall timelines - current macro circumstance has exacerbated supply chain issues across many industries, oxygen being no exception. Not to mention that global oxygen device availability has been a problem for years now - in an April 2020 survey of home medical-equipment suppliers, more than half of reported supply-chain interruptions were for CPAP machines, and 62 percent reported up to a 60-day delay. Couple this industry-specific problem with the semiconductor shortage plaguing every industry (needed for new PAP machines), and you’ve got yourself a uniquely poor time to have a multi-million device recall.
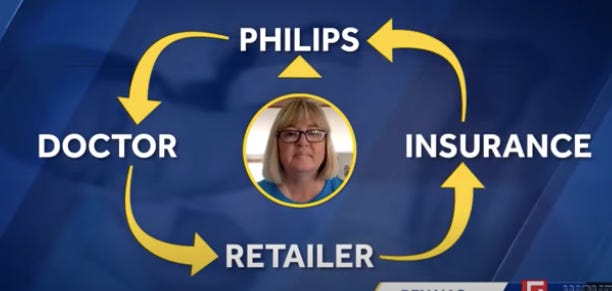
Hw are other stakeholders coming together to fill in the hole that this recall has caused patients? Why, through the tried and true healthcare strategy of Passing the Buck -for those unfamiliar, I’ve borrowed an easy to understand “model” (screenshot from cable news report) of how it works below.
Here’s how it works. First, Philips tells confused patients (if they are even aware that there’s beeb a recall) to follow up with their physician to determine the next steps in their care. Then, the physician points the patient back to Philips, or to the DME supplier they got the initial machine from. The DME supplier says *so sorry, so sorry* that they are not on the hook for the Philips malfunction and can’t replace the equipment. Then, the supplier tells the patient to reach out to their insurance company, who *should* help their patients o find out what the insurance company will cover. Finally, the insurance company tells the patient they can’t cover a new device since the utilization limit is one CPAP every five years - no exceptions for Class I FDA recalls, apparently. But fear not, the insurance company knows just who the patient should talk to, Philips. Healthcare incentives at work, people!
So what can patients actually do?
Well, some patients have decided just to take the risk (maybe buying inexpensive bacterial filters) and live with the recalled device until Philips can send them a new model..sometime in the next twelve months. Other patients, weighing the risks of inhaling carcinogens and living with untreated OSA, have determined that a lower quality of life is better than an increased risk of death via cancer.
And then there are the people who are going to buck up and just pay the $500+ for a new machine from a different supplier. This is frustrating, because there are many people who live on fixed income for which this is a sizable (and in a functional healthcare system, avoidable) expense. When the only options are increased cancer risk, decreased quality of life, or decrease in financial stability, some people are going to pick option 3 (and there’s nothing wrong with that - these are insane choices).
Even when people buck up and pay cash, there’s the issue of overall supply. ResMed, one of the Philip’s largest competitors, told investors on their August earnings call that the company stands to make an additional $350M in 2022 as a result of new business from the Philips recall. That said, they cautioned shareholders that they would have to leave some money on the table despite their good fortune, as their forecasted supply would would not be able to meet the projected new demand. Remember - supply chain shortages were not a new problem for the respiratory DME industry, many patients were already waiting more than 30 days on average for oxygen equipment.
What’s left? Ah yes, Option D - the American Way, a litany of lawsuits. Which is what is happening now (see below)
Philips is now facing at least 40 lawsuits stateside, 32 of which are expected to be centralized after arguments are presented at the end of September in the Eastern District of Pennsylvania. Philips has also raised the ire of a few politicians - see Sen. Blumenthal’s (D-CT) strongly worded letter to Philips US CEO demanding answers for this recall. Unfortunately, even if these lawsuits have merit, and Philips pays out a sizable settlement, this doesn’t really help Philips patients who would like their respiratory treatment without a side of increased cancer risk.
The Long and Short
Recalls happen - it’s just an inevitability. Further, I don’t have the insider baseball knowledge to know if Philips could have avoided the carcinogenic foam issue through better oversight and controls. But to me, it’s the ramifications of the recall that are problematic - we have created a healthcare system where in times of crisis, no player steps up unless they are mandated to do so (front-line workers not included). DME isn’t the only area where healthcare entities pass the buck rather than solve the actual problem, but as a less covered part of the healthcare ecosystem - I figure I’d highlight an egregious example.
My bet: Philips will pay out hundreds of millions of dollars from class action lawsuits and reworking their equipment. Given their paltry options, patients will learn to live with the risks of their Philips equipment, go without their prescribed OSA treatment, or shell out a material amount to get a replacement that they shouldn’t have had to pay for in the first place. ResMed and other competitors will make a ton of money off of crisis demand. Shareholders rejoice.
Will anyone learn anything from this?
I don’t know. I don’t think so. Payors won’t lose anything in the short term for not paying for replacement equipment. Some DME suppliers will be forced to shell out CPAPs free of charge if CMS pushes for it. Policy wonks will have another cautionary tale of the dangers of M&A efficiencies to supply chain resiliency. I have my doubts that mainstream politicians will flex their political muscles to prevent these issues coming up in the future, given the DME. I guess patients they will learn, probably relearn, not to rely too much on the American healthcare system… unless they can pay cash :).
I’ll close with a patient quote I liked which sums this up, from a well-done piece by Kait Sanchez in the Verge. “Respiratory equipment is not like a car that’s faulty,” says Ingrid Tischer. “You can stop driving the car, but you can’t just postpone breathing. So we were given a really ridiculous thing that they called a choice, which was use it or don’t.”
Thanks for reading!
Going to HLTH in Boston in a few weeks? So am I. Would love to meet up if you’re interested in talking Durable, or just want to meet more people in healthcare - which is why I’m going!
Have a technical background? Want to solve (or fail to solve, IDK) a cumbersome, opaque industry with regulatory constraints that Kafka wouldn’t wish on his worst enemy?
Former nurse, case manager, people or physical/occupational therapist? Smooth operator? Seen these issues occur IRL? Want to get people the DME they need easier, and at scale?
Shoot me a message at ed.manzi55@gmail.com. Let’s talk!